XXXI Congresso Nazionale Intergruppo Melanoma Italiano (IMI)
2025: XXXI Congresso Nazionale Intergruppo Melanoma Italiano (IMI)
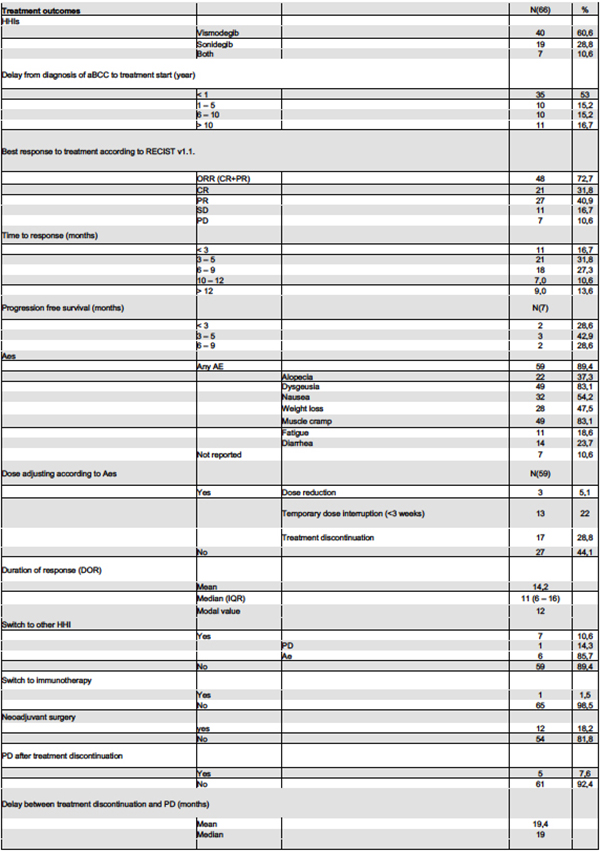
24 | Management of advanced basal cell carcinoma with hedgehog inhibitors: a real-world prospective comparative study
Federico Venturi1|2|3, Biancamaria Zuccaro1, Gabriella Perillo1, Giovanni Cecchi1, Aurora Gaeta4|5, Sara Gandini4, Vincenzo De Giorgi1|6 | 1Section of Dermatology, Department of Health Sciences, University of Florence; 2Oncologic Dermatology Unit, IRCCS Azienda Ospedaliero-Universitaria di Bologna; 3Department of Medical and Surgical Sciences, DIMEC, Alma Mater Studiorum University of Bologna; 4Department of Experimental Oncology, European Institute of Oncology, IRCCS, Milan; 5Department of Statistics and Quantitative Methods, University of Milano-Bicocca; 6Cancer Research "Attilia Pofferi" Foundation, Pistoia, Italy.

Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 11 December 2025
284
Views
0
Downloads