XXXI Congresso Nazionale Intergruppo Melanoma Italiano (IMI)
2025: XXXI Congresso Nazionale Intergruppo Melanoma Italiano (IMI)
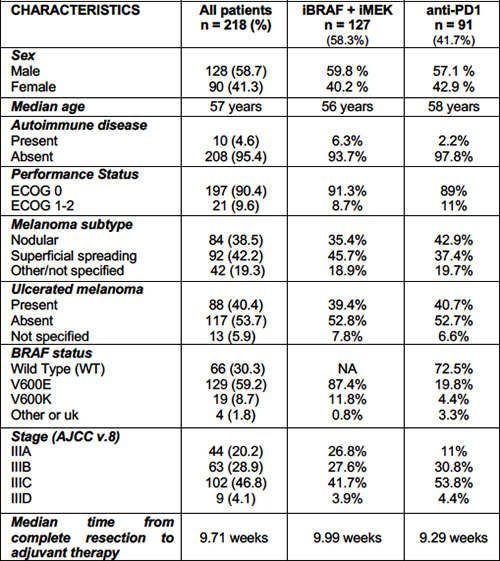
31 | Effectiveness and safety of adjuvant combo-targeted and immunotherapy in melanoma patients
Valentina Salizzato1, Luisa Piccin1, Alessio Fabozzi2, Paolo Del Fiore3, Francesco Cavallin4, Chiara Cacco1|5, Simone Mocellin3|5, Marcodomenico Mazza3, Valentina Guarneri1|5, Jacopo Pigozzo1 | 1Medical Oncology 2, Veneto Institute of Oncology IOV-IRCCS, Padova; 2Medical Oncology 3, Veneto Institute of Oncology IOV-IRCCS, Padova; 3Soft-Tissue, Peritoneum and Melanoma Surgical Oncology, Veneto Institute of Oncology IOV-IRCCS, Padova; 4Independent Statistician, Solagna; 5Department of Surgical, Oncological and Gastroenterological Sciences, University of Padova, Padova, Italy.

Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.
Published: 11 December 2025
276
Views
0
Downloads